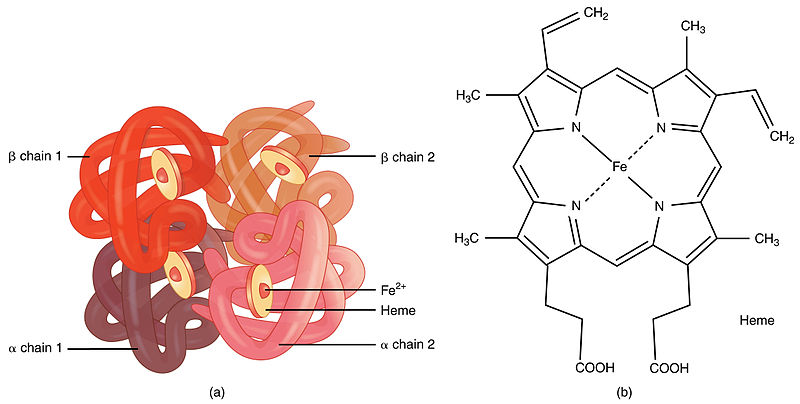
NO is synthesized in multiple cell types and tissues from the substrate L-arginine by NO synthase enzyme systems, which may be constitutive or inducible in response to physiological stimuli. Nitric oxide (NO) has been recognized in the last decade to have important and diverse physiological effects. These strategies include attempts to modify the structure of hemoglobin S to reduce the hydrophobic interactions between hemoglobin β chains or to increase the affinity for oxygen, as well as attempts to decrease the concentration of hemoglobin S by increasing the percentage of fetal hemoglobin or by increasing red cell volume ( 2). A variety of therapeutic strategies for sickle cell disease aimed at reducing hemoglobin S polymerization have been under development in recent years. The P 50 value is inversely related to the hemoglobin affinity for oxygen. This reduced oxygen affinity results in an increase in the partial pressure of oxygen necessary for 50% hemoglobin saturation (P 50). Within the red cell, hemoglobin S has reduced affinity for oxygen, compared with hemoglobin A, because of competition between polymerization and oxygen binding. In sickle cell anemia erythrocytes, deoxygenation of sickle hemoglobin (hemoglobin S) leads to intracellular polymer formation, ultimately distorting the erythrocyte and retarding red cell transit through the microvasculature ( 1). The levels of nitrosylated hemoglobin are too low to affect overall hemoglobin oxygen affinity, but augmented NO transport to the microvasculature seems a promising strategy for improving microvascular perfusion. At baseline, the arterial and venous levels of nitrosylated hemoglobin were not significantly different, but NO inhalation led to a dose-dependent increase in mean nitrosylated hemoglobin, and at the highest dosage, a significant arterial-venous difference emerged. Oxygen affinity, as measured by P 50, did not respond to inhaled NO, either in controls or in individuals with sickle cell disease. We studied the effects of a 2-hour treatment, using varying concentrations of inhaled NO. Also, proposed allosteric mechanisms for hemoglobin, based on S-nitrosation of β-chain cysteine 93, raise the possibilty of altering the pathophysiology of sickle cell disease by inhibiting polymerization or by increasing NO delivery to the tissue.
Nitric oxide (NO) inhalation has been reported to increase the oxygen affinity of sickle cell erythrocytes.
